Prof Franziska Faber
RNA Biology of gram-positive Bacteria (associated research group)
Our research and approach
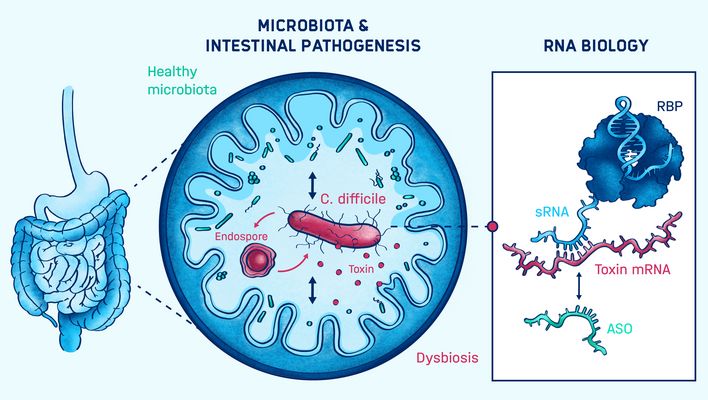
Franziska Faber leads a research group located at the Julius-Maximilians-Universität of Würzburg and associated with HIRI. The group investigates the interplay between the enteric pathogen Clostridioides difficile and the intestinal microbiota. They seek to better understand the RNA-based mechanisms controlling virulence during these interactions. The goal is to leverage this knowledge for the development of novel RNA-based antimicrobials.
Enteric infections and their clinical outcome are often associated with underlying microbial dysbiosis, an imbalance in the microbiota. Clostridioides difficile—a leading cause of antibiotic-associated diarrhea—is a prime example: In healthy individuals, infection with C. difficile results in asymptomatic colonization. However, after antibiotic treatment, the enteric pathogen causes a wide spectrum of diseases. Therefore, a mechanistic understanding of virulence regulation in the context of pathogen-commensal interactions is paramount for the development of novel RNA-based therapeutics.
Franziska Faber’s group addresses this goal through the molecular characterization of virulence regulation, with a focus on post-transcriptional regulation by non-coding RNAs and their cognate RNA-binding proteins. They also identify the microbial and host signals that regulate the virulence gene expression. Furthermore, the Faber group establishes antisense oligomer-based targeting of genes and pathways essential for virulence.
Their work combines technologies from the fields of RNA biology, biochemistry, metabolomics, genetics and microbiology. The overarching goal of the Faber group is to identify and functionally characterize novel RNA-based mechanisms of virulence regulation that can be exploited as therapeutic targets.
Team members

Prof Franziska Faber
Group Leader

Dr Fergal Hamrock
Postdoc

Christopher Birk
PhD Student

Jakob Jeschonneck
PhD Student

Janet Wackenreuter
PhD Student

Johannes Sulzer
PhD Student

Jonathan Scheler
PhD Student

Judith Arbeiter
PhD Student

Sara Giddins
PhD Student

Anna Zweyer
Technical Assistant
Research projects
Graphical Abstract
The production of toxins and the ability to form antibiotic-resistant spores are the major virulence factors in C. difficile. These factors contribute to significant morbidity, mortality, and to high recurrence rates in patients. Their production is tightly regulated by metabolic signals whose abundance and composition throughout the infection cycle is shaped by the gut microbial community.
In our lab, we combine deep RNA-sequencing based approaches with RNA biology, biochemical, genetic and microbiological techniques. We aim to discover and mechanistically characterize non-coding RNAs and their RNA-binding protein partners involved in virulence gene regulation.
We are also interested in how microbial and host signals shape the activities of these regulators. To better understand this cross-talk, we take a bottom-up approach by studying individual pathogen-commensal interactions. To do this, we integrate microbiological, genetic, advanced cell culture and metabolomics approaches.
The overarching goal of our research is to identify regulatory RNAs along with their regulated pathways that may be exploited as therapeutic targets in the future. Towards that goal, we are currently establishing antisense oligomer-based gene silencing in C. difficile focusing on essential genes.
Alumni
Manuela Fuchs, PhD Student & Postdoc • Vanessa Lamm-Schmidt, PhD Student • Tina Lence, Postdoc
Publications
2025
Enabling next-generation anaerobic cultivation through biotechnology to advance functional microbiome research
Clavel T, Faber F, Groussin M, Haller D, Overmann J, Pauvert C, Poyet M, Selkrig J, Stecher B, Typas A, …, Wylensek D, Maier L (2025)
Nature Biotechnology 43 (6): 878-888
The global RNA-binding protein RbpB is a regulator of polysaccharide utilization in Bacteroides thetaiotaomicron
Rüttiger AS, Ryan D, Spiga L, Lamm-Schmidt V, Prezza G, Reichardt S, Langford M, Barquist L, Faber F, Zhu W, Westermann AJ (2025)
Nature Communications 16 (1): 208
Proton-pump inhibitors increase C. difficile infection risk by altering pH rather than by affecting the gut microbiome based on a bioreactor model
Schumacher J, Müller P, Sulzer J, Faber F, Molitor B, Maier L (2025)
Gut Microbes 17 (1): 2519697
An sRNA overexpression library reveals AbnZ as a negative regulator of an essential translocation module in Caulobacter crescentus
Velasco-Gomariz M, Sulzer J, Faber F, Fröhlich KS (2025)
Nucleic Acids Research 53 (1): gkae1139
2024
Formation of the pyruvoyl-dependent proline reductase Prd from Clostridioides difficile requires the maturation enzyme PrdH
Behlendorf C, Diwo M, Neumann-Schaal M, Fuchs M, Körner D, Jänsch L, Faber F, Blankenfeldt W (2024)
PNAS Nexus 3 (7): pgae249
The conserved noncoding RNA ModT coordinates growth and virulence in Clostridioides difficile
Lence T, Sulzer J, Andress K, Gribling-Burrer AS, Lamm-Schmidt V, Barquist L, Smyth RP, Faber F (2024)
PLOS Biology 22 (12): e3002948
2023
Durchfallerreger und die Homöostase der Mikrobiota
Winter S, Faber F, Bäumler A (2023)
BIOspektrum 29 (2): 127-129
A network of small RNAs regulates sporulation initiation in C difficile
Fuchs M, Lamm-Schmidt V, Lence T, Sulzer J, Bublitz A, Wackenreuter J, Gerovac M, Strowig T, Faber F (2023)
The EMBO Journal 42 (12): e112858
2021
RNA landscape of the emerging cancer-associated microbe Fusobacterium nucleatum
Ponath F, Tawk C, Zhu Y, Barquist L, Faber F, Vogel J (2021)
Nature Microbiology 6 (8): 1007-1020
Grad-seq identifies KhpB as a global RNA-binding protein in Clostridioides difficile that regulates toxin production
Lamm-Schmidt V, Fuchs M, Sulzer J, Gerovac M, Hör J, Dersch P, Vogel J, Faber F (2021)
microLife 2: uqab004
Malaria parasite infection compromises colonization resistance to an enteric pathogen by reducing gastric acidity
Walker GT, Yang G, Tsai JY, Rodriguez JL, English BC, Faber F, Souvannaseng L, Butler BP, Tsolis RM (2021)
Science Advances 7 (27): eabd6232
The metabolic footprint of Clostridia and Erysipelotrichia reveals their role in depleting sugar alcohols in the cecum
Tiffany CR, Lee JY, Rogers AWL, Olsan EE, Morales P, Faber F, Bäumler AJ (2021)
Microbiome 9 (1): 174
An RNA-centric global view of Clostridioides difficile reveals broad activity of Hfq in a clinically important gram-positive bacterium
Fuchs M, Lamm-Schmidt V, Sulzer J, Ponath F, Jenniches L, Kirk JA, Fagan RP, Barquist L, Vogel J, Faber F (2021)
PNAS 118 (25): e2103579118
2020
Antibacterial Anacardic Acid Derivatives
Saedtler M, Förtig N, Ohlsen K, Faber F, Masota N, Kowalick K, Holzgrabe U, Meinel L (2020)
ACS Infectious Diseases 6 (7): 1674-1685
2019
Commensal Enterobacteriaceae Protect against Salmonella Colonization through Oxygen Competition
Litvak Y, Mon KKZ, Nguyen H, Chanthavixay G, Liou M, Velazquez EM, Kutter L, Alcantara MA, Byndloss MX, Tiffany CR, …, Zhou H, Bäumler AJ (2019)
Cell Host & Microbe 25 (1): 128-139.e5
Endogenous Enterobacteriaceae underlie variation in susceptibility to Salmonella infection
Velazquez EM, Nguyen H, Heasley KT, Saechao CH, Gil LM, Rogers AWL, Miller BM, Rolston MR, Lopez CA, Litvak Y, …, Byndloss AJ, Bäumler AJ (2019)
Nature Microbiology 4 (6): 1057-1064
Social behaviour and making attachments: a report from the fifth 'Young Microbiologists Symposium on Microbe Signalling, Organisation and Pathogenesis'
An SQ, Ding YC, Faber F, Hobley L, Sá-Pessoa J (2019)
Microbiology 165 (2): 138-145
2018
Genetic Ablation of Butyrate Utilization Attenuates Gastrointestinal Salmonella Disease
Bronner DN, Faber F, Olsan EE, Byndloss MX, Sayed NA, Xu G, Yoo W, Kim D, Ryu S, Lebrilla CB, Bäumler AJ (2018)
Cell Host & Microbe 23 (2): 266-273.e4
2017
Microbiota-activated PPAR-γ signaling inhibits dysbiotic Enterobacteriaceae expansion
Byndloss MX, Olsan EE, Rivera-Chávez F, Tiffany CR, Cevallos SA, Lokken KL, Torres TP, Byndloss AJ, Faber F, Gao Y, …, Lebrilla CB, Bäumler AJ (2017)
Science 357 (6351): 570-575
Colonization resistance: The deconvolution of a complex trait
Olsan EE, Byndloss MX, Faber F, Rivera-Chávez F, Tsolis RM, Bäumler AJ (2017)
The Journal of Biological Chemistry 292 (21): 8577-8581
A Salmonella Regulator Modulates Intestinal Colonization and Use of Phosphonoacetic Acid
Elfenbein JR, Knodler LA, Schaeffer AR, Faber F, Bäumler AJ, Andrews-Polymenis HL (2017)
Frontiers in Cellular and Infection Microbiology 7: 69
Respiration of Microbiota-Derived 1,2-propanediol Drives Salmonella Expansion during Colitis
Faber F, Thiennimitr P, Spiga L, Byndloss MX, Litvak Y, Lawhon S, Andrews-Polymenis HL, Winter SE, Bäumler AJ (2017)
PLOS Pathogens 13 (1): e1006129
2016
Depletion of Butyrate-Producing Clostridia from the Gut Microbiota Drives an Aerobic Luminal Expansion of Salmonella
Rivera-Chávez F, Zhang LF, Faber F, Lopez CA, Byndloss MX, Olsan EE, Xu G, Velazquez EM, Lebrilla CB, Winter SE, Bäumler AJ (2016)
Cell Host & Microbe 19 (4): 443-54
Host-mediated sugar oxidation promotes post-antibiotic pathogen expansion
Faber F, Tran L, Byndloss MX, Lopez CA, Velazquez EM, Kerrinnes T, Nuccio SP, Wangdi T, Fiehn O, Tsolis RM, Bäumler AJ (2016)
Nature 534 (7609): 697-9
2015
Inflammation-associated alterations to the intestinal microbiota reduce colonization resistance against non-typhoidal Salmonella during concurrent malaria parasite infection
Mooney JP, Lokken KL, Byndloss MX, George MD, Velazquez EM, Faber F, Butler BP, Walker GT, Ali MM, Potts R, …, Luckhart S, Tsolis RM (2015)
Scientific Reports 5: 14603
2014
The impact of intestinal inflammation on the nutritional environment of the gut microbiota
Faber F, Bäumler AJ (2014)
Immunology Letters 162 (2 Pt A): 48-53
Salmonella enterica Serovar Typhi conceals the invasion-associated type three secretion system from the innate immune system by gene regulation
Winter SE, Winter MG, Poon V, Keestra AM, Sterzenbach T, Faber F, Costa LF, Cassou F, Costa EA, Alves GE, …, Santos RL, Bäumler AJ (2014)
PLOS Pathogens 10 (7): e1004207
2012
Surface-associated motility, a common trait of clinical isolates of Acinetobacter baumannii, depends on 1,3-diaminopropane
Skiebe E, de Berardinis V, Morczinek P, Kerrinnes T, Faber F, Lepka D, Hammer B, Zimmermann O, Ziesing S, Wichelhaus TA, …, Pfeifer Y, Wilharm G (2012)
International Journal of Medical Microbiology 302 (3): 117-28
2010
A simple and rapid method of bacterial transformation
Wilharm G, Lepka D, Faber F, Hofmann J, Kerrinnes T, Skiebe E (2010)
Journal of Microbiological Methods 80 (2): 215-6
Orbus hercynius gen. nov., sp. nov., isolated from faeces of wild boar, is most closely related to members of the orders 'Enterobacteriales' and Pasteurellales
Volkmann M, Skiebe E, Kerrinnes T, Faber F, Lepka D, Pfeifer Y, Holland G, Bannert N, Wilharm G (2010)
International Journal of Systematic and Evolutionary Microbiology 60 (Pt 11): 2601-2605
2009
CsrA and CsrB are required for the post-transcriptional control of the virulence-associated effector protein AvrA of Salmonella enterica
Kerrinnes T, Zelas ZB, Streckel W, Faber F, Tietze E, Tschäpe H, Yaron S (2009)
International Journal of Medical Microbiology 299 (5): 333-41
2007
Influence of poly(L-lysine) on the structure of dipalmitoylphosphatidylglycerol/water dispersions studied by X-ray scattering
Förster G, Schwieger C, Faber F, Weber T, Blume A (2007)
European Biophysics Journal 36 (4-5): 425-35
2006
Lack of angiotensin II conversion to angiotensin III increases water but not alcohol consumption in aminopeptidase A-deficient mice
Faber F, Gembardt F, Sun X, Mizutani S, Siems WE, Walther T (2006)
Regulatory Peptides 136 (1-3): 130-7