Meningococci are bacteria that can cause life-threatening meningitis and sepsis. These pathogens use a small protein with a large impact: The RNA-binding protein ProQ is involved in the activation of more than 250 bacterial genes.
ProQ ensures that meningococci can better repair their DNA if damaged and it makes them resistant to oxidative stress. Both these factors contribute significantly to the bacteria's pathogenic properties.
This was reported by research groups led by the Würzburg scientists Christoph Schoen from the Julius-Maximilians-Universität (JMU) and Jörg Vogel from the Helmholtz Institute for RNA-based Infection Research (HIRI) in the journal Nature Communications. The HIRI is a joint institution of the Helmholtz Centre for Infection Research (HZI) in Braunschweig and the JMU.
"We were surprised that a comparatively small protein can have such a great influence on bacterial gene regulation," says Christoph Schoen, professor at the Institute of Hygiene and Microbiology at Julius-Maximilians-Universität (JMU) Würzburg in Bavaria, Germany.
ProQ only consists of about 120 amino acids. By comparison, many other proteins are usually made up of several hundred amino acids.
ProQ interacts with 200 RNAs
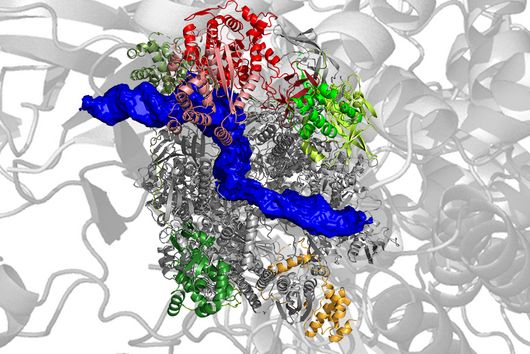
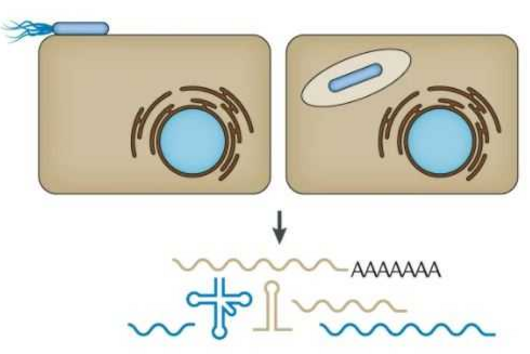
The mini-protein belongs to the group of RNA-binding proteins. RNA molecules play an important role as regulators in many biological processes. They often perform their functions in combination with the binding proteins.
ProQ is a key player in this respect: "In meningococci, the protein interacts with almost 200 different RNA molecules," says Jörg Vogel. "It binds to structured regions of the RNA and thus stabilises its binding partners.“
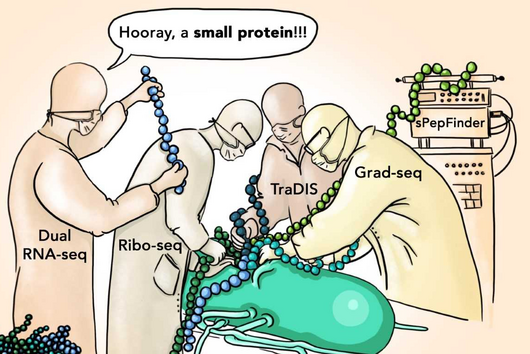
The researchers made the discovery using modern high-throughput processes. These methods were developed in Vogel's group at the Helmholtz Institute for RNA-based Infection Research (HIRI) in Würzburg. Vogel is director of HIRI and heads the JMU Institute for Molecular Infection Biology.
Looking for new drugs against bacteria
The scientists are interested in the processes in bacteria because they hope to find new targets for antibacterial agents. The pathways regulated by RNA and its binding proteins in particular offer a promising field of activity. "We hope to be able to disrupt the binding proteins in their function with small molecules and hence weaken the pathogens," explains Vogel.
Objective: to identify all RNA binding proteins
For two thirds of all RNAs in meningococci, the associated binding proteins have not yet been identified. This raises questions: Is it possible that the majority of RNAs does not need proteins to carry out their regulatory function in bacterial cells? And which processes are actually regulated by RNA-binding proteins?
"This is what we would like to find out – and meningococci are particularly well suited for this task because of their relatively small genome," says Schoen. "Our goal is to systematically identify the entire family of RNA-binding proteins in meningococci using established high-throughput methods."
Original publication
Saskia Bauriedl*, Milan Gerovac*, Nadja Heidrich, Thorsten Bischler, Lars Barquist, Jörg Vogel, Christoph Schoen. The minimal meningococcal ProQ protein has an intrinsic capacity for structure-based global RNA recognition. Nature Communications, 2020, DOI: 10.1038/s41467-020-16650-6
(* these authors contributed equally)
read article
Contact persons
Prof. Dr. Dr. Christoph Schoen
Institute for Hygiene and Microbiology
University of Würzburg
T +49 931 31-46162
cschoen(at)hygiene.uni-wuerzburg.de
Prof. Dr. Jörg Vogel
Helmholtz Institute for RNA-based Infection Research Würzburg
T +49 931 31-82575
joerg.vogel(at)helmholtz-hiri.de
Sponsors
This research work was financially supported by the Bavarian State Ministry of Economic Affairs, Regional Development and Energy, and the Interdisciplinary Centre for Clinical Research (IZKF) Würzburg.