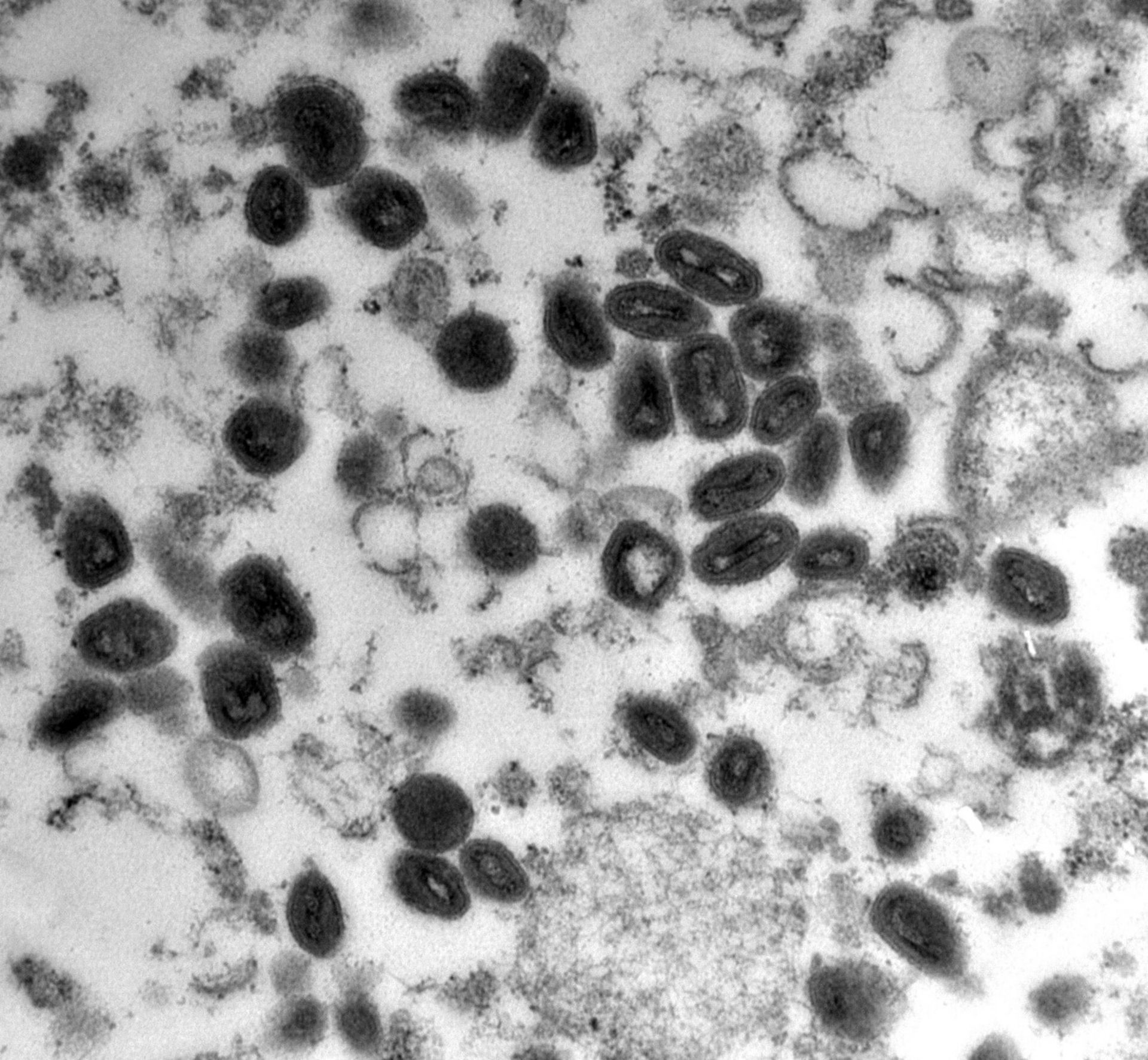
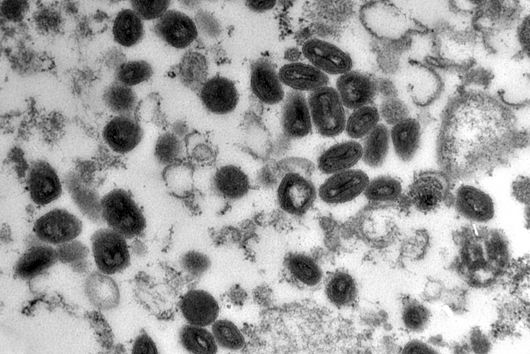
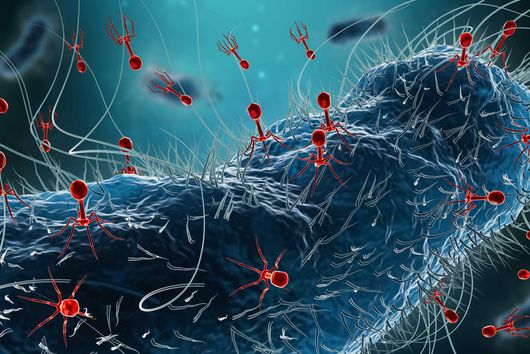
Würzburg, August 1, 2022 – Poxviruses pose a threat to humanity that should be taken seriously, as the current outbreak of monkeypox shows. A research team from the University of Würzburg is now working on the development of new drugs.
The coronavirus pandemic is far from over, and already another virus is causing a stir: “International outbreak of monkeypox” was the cry in the media a few weeks ago, followed by daily headlines along the lines of “first case of monkeypox in Baden-Württemberg, Bavaria, Berlin and so on”. Subsequent reports, like that of Bavaria’s public broadcasting service Bayerischer Rundfunk – “Study: monkeypox pathogen mutates faster than expected” – or that of the German weekly news magazine Der Spiegel – “Doctor says of monkeypox: ‘We have gone past the point in time where the virus could still have been stopped completely’”, did nothing to calm the situation.
And at the latest since the World Health Organisation (WHO) declared the monkeypox outbreak in more than 50 countries an "emergency of international concern" on 23 July, it should be clear to everyone: Viruses can cross species barriers at any time and cause novel diseases, known as zoonoses, in humans. In extreme cases, they can even trigger another pandemic.
Innovative approaches to drug development
Given this context, it seems more than fitting that the University of Würzburg (JMU) is now launching a new research project that deals with this very topic. “A structure-based approach to combat zoonotic poxviruses” is its title. This project, with funding of around EUR 700,000 from the Volkswagen Foundation, is headed by HIRI Affiliate Utz Fischer, Chair of Biochemistry at JMU, and his colleague Clemens Grimm. Also on board is Intana Bioscience GmbH, a biotech company based near Munich with a special interest in the development of new agents. Poxviruses are the focus of this project.
Poxviruses carry a high risk potential
“We know that animals carry many types of viruses in their organism that can pose a threat to humans,” says Utz Fischer. Unfortunately, it is impossible to predict which of them will be the next to make the jump across species barriers. However, it is clear that some viruses have a higher potential than others and are therefore more threatening to humanity, with poxviruses right at the top of this list. The aim of the project is therefore to conceive new approaches to developing novel drugs against pox pathogens. To this end, the scientists are looking for substances that interfere with the viral transcription process and thus prevent the viruses from multiplying.
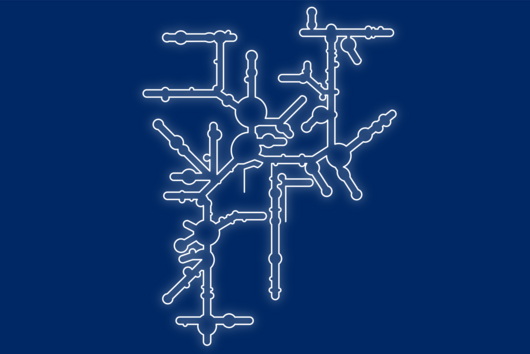
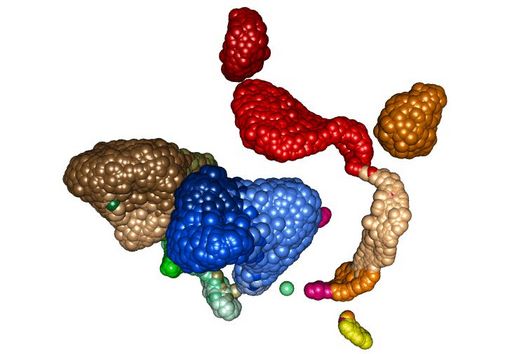
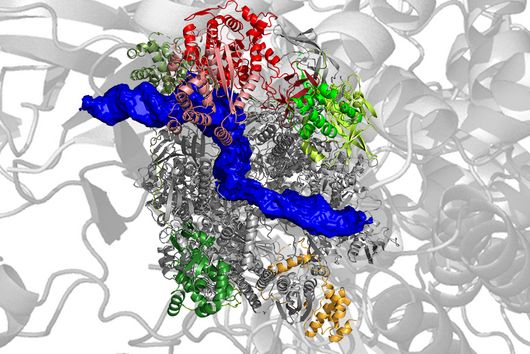
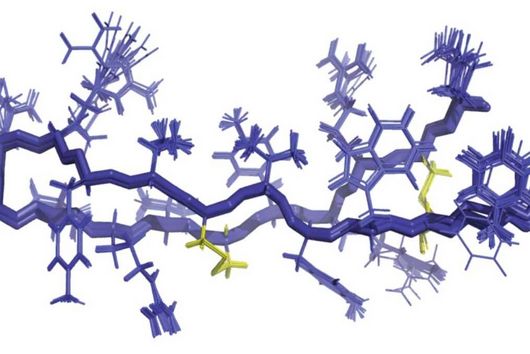
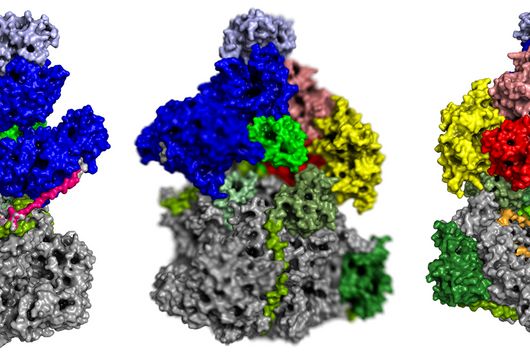
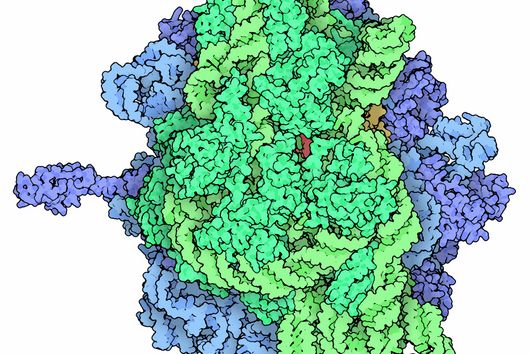
Fischer and his team can draw on findings that they presented to the public last autumn. “We succeeded in visualising the poxviral gene expression machinery at the atomic level,” explains the biochemist. These studies therefore allow to observe how the molecular machinery acts when the virus multiplies. The images show in detail how the players involved operate during the early phase of transcription.
An approach that can also be applied to swine fever
With the search for agents that can inhibit these molecular machines, the team can exploit a special circumstance: While many viruses draw extensively on the biochemical resources of the host cell for their multiplication, poxviruses encode their own molecular machinery in their genome for this purpose. Important components of this machinery are two enzymes: DNA polymerase, which multiplies the viral genes, and RNA polymerase, which transcribes the viral genes into mRNA.
This unique replication strategy offers the opportunity to search for inhibitors of key viral complexes and enzymes that attack at this exact point and thus spare the host cells, ideally making them free from side effects. Should the team succeed in identifying and designing such molecules, they might even kill two birds with one stone: “Because the transcription machineries of Poxviridae and Asfarviridae are highly similar, our research is also likely to be of relevance for the economically highly threatening Asfarvirus-linked swine fever disease,” says Fischer.
Technical progress helps the research
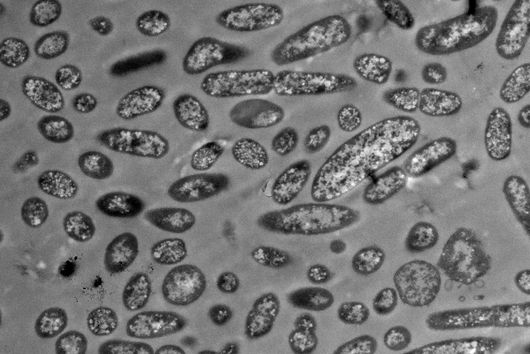
What is most helpful to the scientists in their work are technical advances. One of these is a significant increase in the resolution of cryo-electron microscopic images. With this technology, samples are “flash-frozen” to temperatures of up to minus 180 degrees Celsius. This is what makes it possible to examine biological molecules and complexes in solution and to reconstruct their three-dimensional structure on the scale of atoms.
The JMU has had a suitable electron microscope for many years. “Many medically relevant target molecules are therefore becoming the focus of drug design. We will use this technology alongside established methods to identify molecules that target the special structures of poxviruses and disrupt their multiplication,” explains Clemens Grimm, who carries out structural biology analyses at the department. Indeed, the scientists involved are optimistic that they will succeed over the coming years in defining a number of chemical compounds that can serve as a kind of guiding structure for the subsequent development of a pharmaceutical product.
Poxviruses are a threat for several reasons
It is, of course, a coincidence that the research team is starting its work at the same time as this outbreak of monkeypox – the application was written months ago. But what is not a coincidence is that the scientists are focusing on poxviruses. After all, these pose a potential threat to humanity for various reasons. For one thing, there are currently very few antiviral drugs available, and these often display very limited potency. In addition, while there is effective protection against pox infection in the form of vaccination, after the World Health Organization (WHO) declared smallpox eradicated in 1980, the corresponding vaccination campaigns were terminated in the ensuing years. Since then, herd immunity to poxviruses has been noticeably dwindling in humans. It is no wonder that a recent study ranks the monkeypox virus as one of the most threatening viruses because of the risk of infecting humans, adapting to its new host through mutations and then spreading exponentially.
Text: JMU Press office
Contact
Prof. Dr Utz Fischer, Department of Biochemistry, University of Würzburg (JMU), T: +49 931 31-84029, utz.fischer(at)biozentrum.uni-wuerzburg.de